
Our research integrates molecular or biological catalysis with nanomaterials to generate solar fuels. We utilise the excellent light absorption properties of nanomaterials to generate charge-separated states that can drive redox processes. Immobilised molecular and biological catalysts allow us to control the selectivity of these redox reactions to produce chemical fuels such as H2 or CO from water and waste. See below for examples of our recent work.
Solar-driven H2 Generation from Waste
The conversion of waste materials into fuels is an attractive approach to simultaneously produce fuels and mitigate pollution. We are developing photocatalysts to drive this process with solar energy.

Lignocellulosic biomass is the most abundant raw material on earth, but its utilisation for fuel production is limited to energy-intensive processes due to low solubility and reactivity at ambient conditions (Angewandte 2018). We demonstrate one of the first examples of direct photochemical conversion of unprocessed waste biomass into clean hydrogen fuel. We adapted concepts of solar fuels research for biomass valorisation by replacing commonly used sacrificial electron donors with waste. Through protection of CdS quantum dots with a CdOx layer to overcome the well-documented photocorrosion of CdS, we developed an efficient and robust photocatalyst. This visible-light responsive system produces H2 fuel from various types of unprocessed lignocellulose such as wood, grass and waste paper at ambient conditions using sunlight as the sole energy input (Nat Energy 2017).

We have extended this approach to the photochemical generation of hydrogen fuel from plastics (Energy Environ Sci 2018). Plastic waste has become a growing environmental challenge of global dimensions that urgently requires new mitigation technologies. Using our CdS/CdOx photocatalyst, we can generate H2 fuel from common plastics such as PET, PLA and PU upon solar irradiation. During this process, the plastics are degraded to useful organic compounds, e.g. photochemical treatment of PET generates terephthalic acid, one of the precursors needed to produce new PET. Remarkably, we can use real-world plastic waste cut from an old dirty water bottle without lowering the performance of our catalyst. We aim for this low-energy process to become a complement to conventional recycling, targeting mixed and contaminated waste that is currently challenging to recycle.
 Together with Prof. Pitchaimuthu at Heriot-Watt, we subsequently translated this concept into a photoelectrochemical process, in which we simultaneously produce H₂ and remove heavy metals from contaminated water using a CdS/TiO₂ photoanode (Sustainable Energy Fuels 2021). Similarly, using a WO₃/BiVO₄ photoanode, surfactants or pharmaceuticals can be degraded while generating H₂ at the same time (J Environ Chem Eng 2023, Ind Eng Chem Res 2023). Recently, together with Prof. Rauf at Lahore University of Management Sciences, we developed a related electrochemical process in which formic acid is produced in both half-reactions—on the one hand through oxidation of plastic waste at CuO@Ni(OH)₂ electrodes, and on the other through CO₂ reduction at Pb–SnO electrodes (ACS Sus Chem Eng 2024).
Together with Prof. Pitchaimuthu at Heriot-Watt, we subsequently translated this concept into a photoelectrochemical process, in which we simultaneously produce H₂ and remove heavy metals from contaminated water using a CdS/TiO₂ photoanode (Sustainable Energy Fuels 2021). Similarly, using a WO₃/BiVO₄ photoanode, surfactants or pharmaceuticals can be degraded while generating H₂ at the same time (J Environ Chem Eng 2023, Ind Eng Chem Res 2023). Recently, together with Prof. Rauf at Lahore University of Management Sciences, we developed a related electrochemical process in which formic acid is produced in both half-reactions—on the one hand through oxidation of plastic waste at CuO@Ni(OH)₂ electrodes, and on the other through CO₂ reduction at Pb–SnO electrodes (ACS Sus Chem Eng 2024).
Selective reduction of aqueous CO2 using immobilised molecular catalysts

Direct solar-driven conversion of H2O and CO2 into feedstock chemicals is a promising strategy to mitigate greenhouse gas emissions and simultaneously store solar energy in chemical form. We have developed entirely precious metal-free photocatalysts for the selective reduction of CO2 to CO in water, based on a hybrid design that combines the photo-physical properties of semiconductor nanocrystals with the selectivity of a well-defined molecular base-metal catalyst. We developed a series of nickel terpyridine complexes with promising electrocatalytic activity for selective CO2 reduction at low overpotential in organic media (JACS 2017). Designing suitable surface anchors allowed us to attach these catalysts to CdS quantum dots to drive CO2 reduction with visible light. Immobilisation enables the Ni catalysts to operate in pH neutral aqueous solution without compromising their selectivity. Under visible light, this precious-metal free CdS-[Ni] hybrid catalyst gave CO with >90% selectivity.

A second-generation photocatalyst based on ZnSe nanocrystals and an immobilised Ni(cyclam) catalyst shows a much higher activity and stability (Chem Sci 2018). Selectivity tuning was achieved by targeted inhibition of hydrogen evolution from the ZnSe particles through surface modification. We have translated these suspension systems to a photoelectrode, by immobilising a Co terpyridine catalyst on a mesoporous Si-TiO2 photocathode for photoelectrochemical CO2 reduction (Nat Catal 2019) and onto a dark cabron nanotube-based electrode (JACS 2017).
 In our most recent work, we exploit hydrophobic interactions of a perfluoroalkylated Co(terpy-RF) complex for catalyst anchoring. At the gas/liquid/solid three-phase boundary of a gas diffusion electrode, these interactions lead to self-organization of the catalyst and the formation of hydrophobic pockets, which shifts the product selectivity from CO toward methane and significantly increases the activity. Using operando Raman spectroscopy, we were able to show that the terpyridine ligand functions as a transporter for protons from the aqueous phase into the hydrophobic pockets (Nat Chem 2025).
In our most recent work, we exploit hydrophobic interactions of a perfluoroalkylated Co(terpy-RF) complex for catalyst anchoring. At the gas/liquid/solid three-phase boundary of a gas diffusion electrode, these interactions lead to self-organization of the catalyst and the formation of hydrophobic pockets, which shifts the product selectivity from CO toward methane and significantly increases the activity. Using operando Raman spectroscopy, we were able to show that the terpyridine ligand functions as a transporter for protons from the aqueous phase into the hydrophobic pockets (Nat Chem 2025).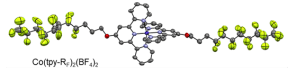
Cooperative solvent design for solar fuel production
A major paradigm in catalysis research is that for improving catalytic activity, new catalysts with modified structures are needed. We pursue the opposite approach by seeking to influence the properties of existing catalysts solely through changes in the solvent.
Photocatalytic processes are typically air-sensitive, because O₂ quenches excited states and inhibits catalytically active centers. We therefore investigate novel solvents that can create an adaptive protective environment capable of maintaining catalytic activity even under challenging conditions. Using a redox probe, we have developed smart solvents that extend the lifetime of air-sensitive species by up to two orders of magnitude (Chem 2020).

Building on our experience with O₂-tolerant photocatalytic H₂ evolution (Chem Eur J 2018), we apply this approach to impart O₂ tolerance to otherwise O₂-intolerant photocatalysts through the use of tailored solvents. With this approach, C₃N₄ retains over 80% of its activity and quantum yield for H₂ production in air when used in a smart solvent, whereas its activity in water is almost completely suppressed by air (Energy Environ Sci 2021).
 Biophotocatalysis is a fascinating field that combines synthetic materials with biologial catalysts into highly active and selective catalysts, often without the use of precious metals (Trends Chem 2025). Together with our collaborators Dr. Cavazza and Dr. Le Goff in Grenoble, we have extended the concept of smart solvents to O₂-sensitive enzymes even enables the use of [NiFeSe]-hydrogenase for photocatalytic H₂ production in air (Angewandte 2023). This not only greatly simplifies practical application but also significantly enhances the longevity of the fragile enzyme.
Biophotocatalysis is a fascinating field that combines synthetic materials with biologial catalysts into highly active and selective catalysts, often without the use of precious metals (Trends Chem 2025). Together with our collaborators Dr. Cavazza and Dr. Le Goff in Grenoble, we have extended the concept of smart solvents to O₂-sensitive enzymes even enables the use of [NiFeSe]-hydrogenase for photocatalytic H₂ production in air (Angewandte 2023). This not only greatly simplifies practical application but also significantly enhances the longevity of the fragile enzyme.
